The Beer-Lambert Law is an essential principle in spectroscopy. It describes the correlation between light absorbance in a specimen and the concentration of absorbing species in the said specimen.
Despite its broad usefulness, various limitations can greatly impact the law’s accuracy and suitability in different scenarios. It’s crucial to highlight these major limitations of the Beer-Lambert Law.
Also, even though the Beer-Lambert Law has its restrictions, there are instances and scenarios where these constraints do not significantly impact its accuracy and applicability.
You must consider these limitations when applying the Beer-Lambert Law to your spectroscopic analyses.
12 Limitations of Beer-Lambert Laws
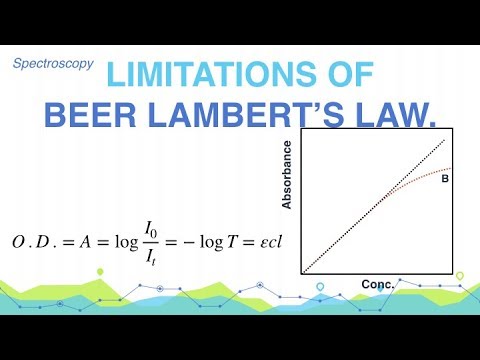
Limitations of Beer-Lambert Laws can arise from different factors, and in this article, we will show you twelve instances where they are applicable. In instances like:
- Heterogeneity
- Nonlinear concentration ranges
- Photochemical reactions
- Temperature dependence
- Path length inconsistencies
- Instrumental limitations
- Interference from other species
- Sample transparency
- Volume variations
- Scattering, reflection effects, etc.
12 Limitations of Beer-Lambert Laws
1. Heterogeneity Assumption
The law assumes that the sample is a homogeneous solution with a uniform concentration of the absorbing species.
However, some samples may not always conform to this condition, leading to inaccuracies in the calculated concentrations.
Most times, heterogeneous mixtures or samples with non-uniform concentration distributions can cause deviations from the ideal Beer-Lambert behavior.
2. Linearity Limit
The law assumes that the absorption of light is linearly related to the concentration of the absorbing species. This linearity is generally valid for low to moderate concentrations.
However, at higher concentrations, the assumption breaks down due to concentration quenching or inner filter effects.
These phenomena can lead to a significant overestimation or underestimation of the actual concentrations.
3. Photochemical Reactions
When light falls on the absorbing species, the Beer-Lambert Law assumes that no photochemical reactions take place. And there are instances where the absorbed light energy triggers chemical changes in the specimen.
And because of this, one can suggest that these photochemical reactions should be considered by researchers for more studies. Particularly with delicate or unstable substances instead.
4. Temperature Dependence
The absorption of light, according to Beer-Lambert Law, is supposed to be unaffected by temperature related changes in sample properties like viscosity, density, or refractive index.
However, these properties do have an impact on absorption and should be considered when interpreting outcomes.
Also, an accurate concentration measurement can only be achieved by accounting for some temperature induced alterations.
5. Assumption of Light Path Length
The Beer-Lambert Law is based on the assumption that the path traveled by light through the sample remains consistent and uniform.
However, practical situations may lead to deviations from this assumption due to factors like uneven distribution of the sample, scattering, or cloudiness.
And these changes could cause inaccurate absorption readings, impacting the concentrations calculated.
6. Instrument Constraints
The derivation of the Beer-Lambert Law also presumes ideal conditions where both the light source and detector have no imperfections or noise.
But in reality, experimental setups often face limitations in the quality of the light source, sensitivity of the detector, or drift in instrumentation.
These constraints can introduce inaccuracies in absorbance measurements, subsequently affecting the precision of concentration determinations.
7. Presence of Interfering Species
The Beer-Lambert Law states that light absorption is solely caused by the specific absorbing species of interest.
However, in some complex mixtures, other absorbing species may interfere, and this can lead to an incorrect concentration reading.
Furthermore, researchers must try to confirm that the absorption only comes from the species of interest.
They can also check to consider the presence of other absorbing species during their calculations to avoid these limitations of Beer-Lambert laws.
8. Transparency of Samples
The law also states that the sample allows light used for measurement to pass through it. Although, some samples may be opaque or translucent, this results in inaccurate measurement, absorption and concentration determination. And to solve this, researchers should verify that the sample is transparent enough for precise measurements.
9. Variability in Sample Volume
The Beer-Lambert law also assumes that the sample volume will remain in a constant state during measurement.
However, it might still experience some changes in sample volume due to factors like evaporation, sedimentation, or diffusion.
And these insignificant changes can affect the absorbance measurement, which will lead to an inaccurate concentration calculation.
10. Wavelength Consideration
The law assumes that the light source emits light at a single, specific wavelength. However, practical light sources often emit a wide range of wavelengths.
This sort of inconsistency can lead to light absorption by various sample species and also result in a deviation from ideal Beer-Lambert behavior.
With each passing day, researchers need to start ensuring that the light source is monochromatic. Or better yet, they can ensure they adjust the influence of multiple wavelengths during any measurement.
11. Uniform Sample Preparation
The Beer-Lambert Law is also based on the assumption that samples are consistently prepared and managed proportionally.
However, there have been alterations in some sample preparations, such as variations in pH, temperature, or the presence of impurities.
And these discrepancies can affect the absorbance measurements, which always leads to these inaccurate concentration determinations.
Hence, advanced research can be done to ensure that sample preparation remains consistent and controlled to minimize these effects.
Impact of Scattering and Reflection
The law assumes that light passes through the sample without significant scattering or reflection. However, real samples may experience scattering and reflection due to factors like particle size, sample cloudiness, or surface roughness.
These phenomena can influence absorbance measurements, resulting in inaccurate concentration calculations.
Researchers need to account for scattering and reflection in their measurements to achieve reliable results.
Conclusion
Conclusively, note that despite its significance in spectroscopy, it is crucial to recognize and address the limitations of Beer-Lambert Law when utilizing it for practical samples.
And by acknowledging these limitations and learning how to implement some necessary corrective actions, a lot of changes can be made.
Also, scientists can enhance the dependability and precision of their concentration determinations. It is very important to understand that these constraints might be challenging to eradicate entirely.
And so, researchers should aim to reduce their influence on the final outcomes afterward.


